


Computational Nanochemistry @ UOIT
Fedor
Naumkin
Associate
Professor of Chemistry

Address:
Faculty
of Science
UOIT
Oshawa,
ON L1H 7K4
Canada
Phone:
+1-905-721-8668
x2942
E-mail:
M.
Sc. Moscow State Engineering Physics
Institute
Ph.
D. General Physics Institute (Russian
Academy of Sciences)
Teaching
Fall:
CHEM
2010U Structure and Bonding
CHEM
3140U Physical Chemistry for Biosciences
Winter:
CHEM
4060U Quantum Chemistry and Spectroscopy
CHEM
2040U Thermodynamics and Kinetics
Research
interests
The
expertise is in the domains of Theoretical / Computational
Chemistry & Chemical Physics,
with
a particular focus on interdisciplinary Nanoscience (studies of nanometer-sized
systems).
The
objects of interest are generally specified as polyatomic systems (atomic and
molecular
complexes
and clusters, atoms and molecule on surfaces, interfaces at molecular level).
Present
directions of research focus on core-shell cluster nano-systems with molecular
species inside
metal-atom
cages (interacting non- and covalently), and on intermolecular junctions
mediated by
ion-pair
links or metal atoms. Other interests include multi-electronic-state model
potentials,
determination
and efficient representation of multi-dimensional intermolecular potential
surfaces.
The
main aims are to predict new stable structures and compositions, to investigate
relationships
between
various properties, and ultimately to design systems with desirable
characteristics.
Potential
practical applications include development of new materials with unique
properties,
novel catalytic agents, elements of molecular electronics and machinery,
molecular
storage and delivery, energy storage at
molecular level.
Research
projects
(1) Clusters - doped and mixed metal-based systems
A
new family of molecule-doped metal clusters is investigated (unlike previously
studied atom-doped systems),
allowing new dimensions in
structure and property design and enabling creation of unique core-shell
species (filled cages);
current
focus is on carbon-, hydrogen- and hydrocarbon-doped metal species.
(2) Charge-transfer intermolecular insertion complexes
Novel
types of complexes are discovered, with molecules trapped between
atomic/molecular ion-pairs,
exhibiting
unusual bond patterns and unique characteristics such as hyper-valence of 2nd-row
elements (carbon) and huge dipole moments,
allowing energy storage in
metastable states and use of molecules in a strong electric field;
current
focus is on compounds of main group metals with halo-hydrocarbons.
(3) Metal-organic molecular interfaces
Increased
stabilization and charge/excitation-induced geometry variation in sandwich
complexes of metal atoms with unsaturated molecules is explored,
enabling
construction of highly stable metal-mediated junctions and 1D polymeric chains
to 3D frameworks with novel properties,
and offering new options for
molecular structure alterations;
current
focus is on main group metal atoms with hydrocarbons.
Research
assistants
Postdoctoral
fellows:
Former:
Ben
Irving
Ramachandran
Chelat
Students
Former:
Stephen
Kerr
Bryce
Cochrane
Kayla
Fisher
Fazil
Momand
Gurpaul
Kochhar
Phillip
McNelles
Hobart
Leung
Selected
recent publications
B. Cochrane and F. Y. Naumkin,
Reshaping and linking of molecules in ion-pair traps.
Chem. Phys. Lett. 643 (2016) 137-141.
[Abstract]
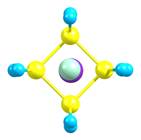
B. J. Irving and F.
Y. Naumkin,
A density functional
investigation of structure-property evolution in the tetrakis-hexahedral C4Al14
nanocluster.
J. Chem. Phys. 141
(2014) 131102. [Abstract] [Paper]

F. Y. Naumkin and K. Fisher, Small metal–organic molecular sandwiches:
Versatile units for induced structure manipulation.
Chem. Phys. Lett. 590 (2013) 52-57.
[Abstract] [Paper]

F. Y. Naumkin and D. J. Wales,
Hydrogen trapped in Ben
cluster cages: The atomic encapsulation option.
Chem. Phys. Lett. 545 (2012) 44-55. [Abstract] [Paper]
